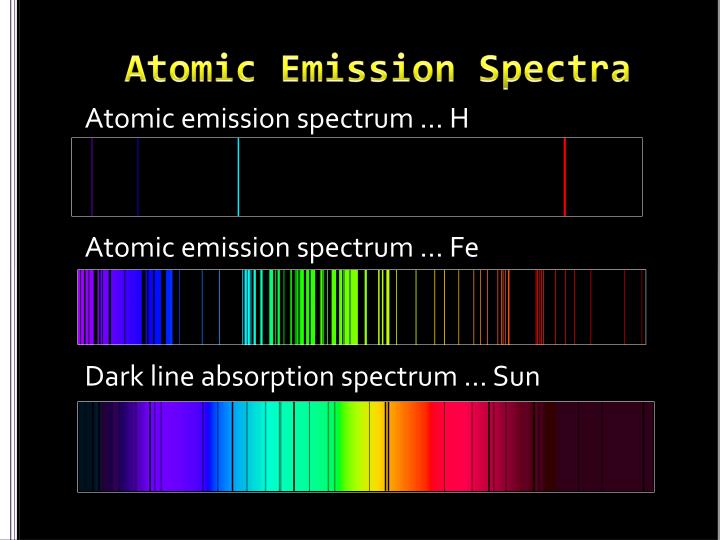 
from Wikipedia.īy contrast, if the detector sees photons emitted directly from a glowing gas, then the detector often sees photons emitted in a narrow frequency range by quantum emission processes in atoms in the hot gas, resulting in an emission line. When an excited atom returns to the ground level, it emits radiation in a discrete wavelength. When an electron gets excited from one energy level to another, it either emits or absorbs light of a specific wavelength. Atomic emission spectrum the variety of frequencies of electromagnetic radiation (light) seen through a spectroscope that are emitted by electrons as they. Atomic emission spectroscopy (AES) is an analytical technique used for the quantification of metal atoms by measuring the intensity of light emitted by the atoms in excited states. spectra, a linear calibration curve of the absorbance versus concentration. Dips are present at the Fraunhofer line wavelengths. The spectrum of the electromagnetic radiation emitted or absorbed by an electron during transitions between different energy levels within an atom. The yellow glow is the fluorescence emission at 560 nm. Emission spectrum definition, the spectrum formed by electromagnetic radiations emitted by a given source, characteristic of the source and the type of excitation inducing the radiations. Free atoms of the material are produced when the flame heat evaporates the solvent and breaks the chemical bonds of the analyte. chemical species under investigation can be derived from X-ray emission spectra.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
